Uses And Side Effects Of Brimonidine

What is brimonidine?
- Pupil dilation
- Decreased saliva production
- Increased heart rate
- Slowing down the functions of the organs of the digestive system
How is brimonidine used?
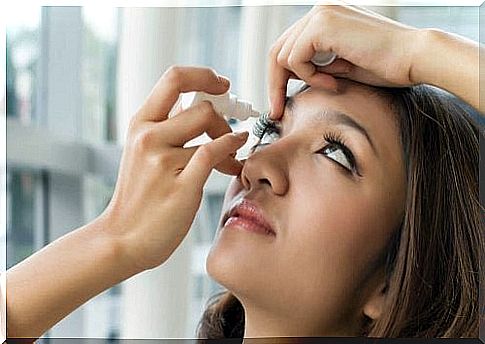
Brimonidine is available as eye drops. In adults and elderly patients, the recommended dose is one drop in the affected eye twice a day, with an interval of about 12 hours between administrations.
Instructions and recommendations for use
Remember that you should not use this medicine if the lid seal is broken. It is important to wash your hands thoroughly before opening the bottle with brimonidine.
- First, he stretches his neck, leans his head back, and looks at the ceiling.
- Then pull very lightly on the bottom of the eye.
- Turn the solution bottle upside down and press on it until a drop of liquid flows into the affected eye.
- Close your eyes.
- For optimal use and to avoid possible systemic absorption, specialists recommend pressing on the tear bag for one minute after applying each drop. In addition, if the patient uses more than one ophthalmic medicine, each should be administered at an interval of about 15 minutes from the next.
- Once the bottle of brimonidine is opened, the medicine should be used within a maximum of 28 days.
Uses and side effects of brimonidine
The main side effects of brimonidine are:
- Dry mouth
- Ocular hyperemia
- Burning sensation and itchy eyes
- Headache
- Insomnia
However, these side effects of brimonidine are usually transient and do not require discontinuation of brimonidine.
Uncommon side effects include:
- Eye fatigue
- Photophobia or sensitivity to light
- Swelling of the eyelids
- Asthenia
- Heart palpitations or arrhythmias (rare)

If you experience the following side effects, see a doctor immediately:
- Irritation
- Difficulty breathing
- Faint
- Dizziness
- Feeling nauseous
Contraindications to brimonidine
Brimonidine treatment is contraindicated in case of:
- The patient’s age is less than 2 years.
Can I use brimonidine if I am pregnant?
Some animal studies show that brimonidine does not cause teratogenic effects. However, studies in rabbits, which detected higher levels of brimonidine than those used during treatment in humans, showed a reduced postnatal growth of the child.
The safety of this medicine during pregnancy in humans has not been established. Therefore, the use of brimonidine is not recommended during pregnancy.
Doctors will only consider using brimonidine if the potential benefit to the mother justifies the potential risk to the fetus. In addition, the use of brimonidine during breast-feeding is not recommended.